
Glaucoma treatment has traditionally relied on daily eye drops. While these can be effective, many patients find it difficult to use them consistently. A new treatment option, iDose® TR (travoprost intracameral implant), offers an alternative: continuous, long-term medication delivery directly inside the eye without the need for daily drops.
Approved by the U.S. Food and Drug Administration (FDA) in December 2023, iDose TR represents a significant advancement in the treatment of open-angle glaucoma and ocular hypertension.
What Is Glaucoma?
Glaucoma is a group of progressive eye diseases characterized by damage to the optic nerve, most commonly associated with elevated intraocular pressure (IOP). It is one of the leading causes of irreversible blindness worldwide.
Early-stage glaucoma typically has no noticeable symptoms, which is why it is often referred to as the “silent thief of sight.” Without treatment, vision loss is permanent. However, early diagnosis and consistent IOP control can significantly slow or prevent progression.
Traditional Eye Drop Therapy
Eye drops are often the first step in treatment, but they can be hard to keep up with. Common challenges include forgetting to take them every day, difficulty putting drops in the eye, burning or irritation from preservatives, and the ongoing cost of medications. Missing doses due to these and other challenges can make it harder to keep eye pressure under control and potentially lead to vision loss.
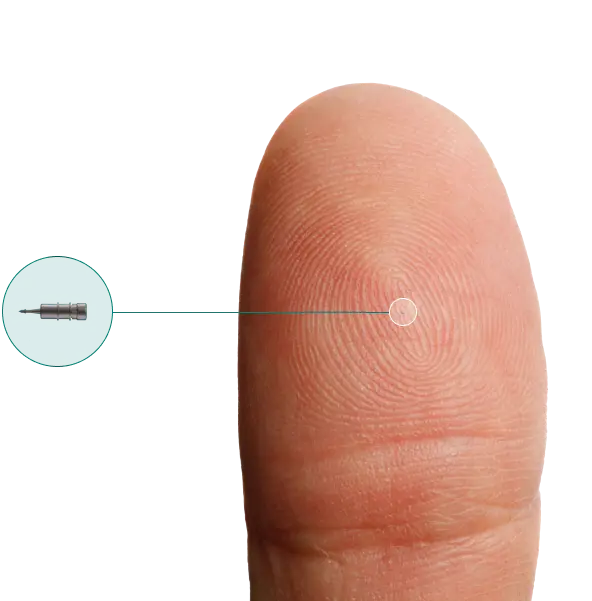
What Is iDose® TR?
iDose TR is a tiny implant that slowly releases glaucoma medication inside the eye over time. About the size of a grain of rice, iDose TR is made of medical-grade titanium, and it delivers a controlled release of medication continuously, day and night. Because the medication is delivered directly inside the eye, it avoids the surface irritation that eye drops can sometimes cause.
How the Procedure Works
The iDose TR implantation is a brief, minimally invasive outpatient procedure:
- The eye is numbed with anesthetic drops
- A small corneal incision is made
- The implant is inserted and secured within the eye’s natural drainage system
- The procedure is typically completed in minutes
It can be performed as a standalone procedure or in combination with cataract surgery.
Does it Work?
Clinical studies have shown encouraging results:
- About 80% of patients did not need daily eye drops at 12 months
- Eye pressure was reduced to levels similar to traditional medications
- Many patients maintained good pressure control for several years
Results can vary from person to person, but for many, this treatment significantly reduces the burden of daily medication.
Is It Safe?
iDose TR has been shown to be generally safe and well-tolerated. Some patients may experience mild, temporary side effects, including:
- Eye redness or irritation
- Mild inflammation
- Temporary increase in eye pressure
- Dry eye symptoms
Your doctor will monitor your eye health over time to ensure everything remains stable.
Important: If you have an iDose TR implant, you should inform your healthcare providers before undergoing an MRI scan, as the implant has specific MRI conditional guidelines.
Who Is a Candidate?
iDose TR is FDA-approved for adults with open-angle glaucoma and ocular hypertension
You may be a good candidate for iDose if you:
- Have trouble remembering or using eye drops
- Experience irritation from glaucoma medications
- Want to simplify your treatment routine
- Are planning to have cataract surgery
- Have responded well to similar medications in the past
A full eye exam is needed to determine if this option is right for you.
What Happens Over Time?
The implant is designed for long-term drug delivery, with clinical studies demonstrating multi-year effectiveness. However:
- The implant generally lasts for several years, but the duration varies between individuals
- Some patients may need another implant later
- Regular follow-up visits are still important
Questions to Ask Your Eye Doctor
If you’re considering iDose TR, ask:
- Am I a candidate for this treatment?
- Can it be combined with cataract surgery?
- Will I still need eye drops afterward?
- How long will the implant last in my case?
- What are the risks and follow-up requirements?
- Is this covered by my insurance or Medicare?
Why Choose Northwest Eye Surgeons?
At Northwest Eye Surgeons, our team of fellowship-trained ophthalmologists is dedicated to staying at the forefront of vision care, bringing the latest evidence-based treatments to our patients in the Seattle area and beyond. We are committed to offering advanced, minimally invasive options like the iDose TR because we believe that protecting your vision should be as seamless and comfortable as possible.
When you choose Northwest Eye Surgeons, you are choosing a practice that takes the time to understand your unique situation, answer your questions thoroughly, and build a glaucoma management plan that fits your life.
Take the Next Step
If you are living with glaucoma or ocular hypertension and are interested in reducing your dependence on daily eye drops, iDose TR may be an option.
Call Northwest Eye Surgeons today to learn whether this treatment is right for you!
Glaukos Corporation. iDose® TR Product Information and Safety Data.
Singh IP. A Deep Dive on the iDose® TR Phase 2b Trial. Glaucoma Today.
Harp MD. Clinical updates on iDose platform. Ophthalmology Times.
U.S. Food and Drug Administration (FDA). iDose TR Approval Summary (2023).
Disclaimer: This blog post is intended for educational purposes only and does not constitute medical advice. The iDose TR is indicated for the reduction of intraocular pressure in patients with open-angle glaucoma or ocular hypertension. As with any medical procedure, individual results may vary. Please consult with your Northwest Eye Surgeons physician to determine whether iDose TR is appropriate for your specific situation. iDose TR is a registered trademark of Glaukos Corporation.



